
Accelerating MedTech Concept Systems Approval Manufacturing Scale
Your end-to-end integrated partner for program acceleration
Accelerating Medtech. Delivering Markets.
Complexity, Solved. Innovation, Accelerated
The orthopedic landscape is evolving rapidly. Your path to market shouldn’t slow you down. EXALTA is the next-generation medtech solutions provider, fusing deep contract manufacturing expertise with integrated OEM system development to bring your vision to life, faster and smarter.

integrated OEM SOLUTIONS
The Fast Track to Market Leadership
Your head start for innovation. Access our library of patented, regulatory-cleared “technology bricks” and comprehensive platforms (spanning Spine, Extremities, Trauma, Recon, and Cardiothoracic).
- Leverage turnkey 510(k)- and EU MDR-cleared systems.
- Utilize validated design data and documentation templates.
- Co-develop differentiated products to rapidly fill portfolio gaps.
The
Difference.

Manufacturing Solutions
Precision, Quality, and Global Scale.
The backbone of orthopedic excellence. We deliver mission-critical precision instrumentation and implants with uncompromising quality, optimized for the ASC environment and seamless integration with robotics.
- Global manufacturing footprint.
- Advanced engineering expertise.
- Nearshoring options for supply chain resilience.
THE RESULT
DECREASE
0%
TIME TO MARKET
DECREASE
0%
IN COST
Traditional CMOs manufacture. EXALTA co-develops.
Speed. Agility. Scale. In One Partner.
We integrate design, engineering, regulatory, manufacturing, and post-market surveillance inside a single platform, so you move from idea to income without piecemealing suppliers.
THE RIGHT PARTNER
Proprietary design capabilities
We combine cutting-edge innovation with medical-grade engineering excellence, delivering differentiated products that capture market share while maintaining IP protection and competitive advantage.
DESIGN


YOU'RE IN GOOD HAND
Supporting Documentation With Unique Options
Comprehensive technical file development and maintenance by regulatory experts ensures first-time approval success and accelerated market entry.
SPEED
ISO 13485
LEGAL MANUFACTURER QMS certified
We are Certified legal manufacturer status with established QMS infrastructure eliminates setup delays and provides immediate production capability at scale.
TRUST


shorten approval
Regulatory Registrations FDA & CE-MDR
We combine cutting-edge innovation with medical-grade engineering excellence, delivering differentiated products that capture market share while maintaining IP protection and competitive advantage.
GLOBAL
TIME TO MARKET
DESIGN FOR MANUFACTURING REDUCED BY UP TO 40%
Advanced DFM optimization transforms innovative concepts into manufacturable products while reducing production costs and time-to-market by up to 40%.
DFM


Consistent quality
manufacturing with Scalability
State-of-the-art production facilities with validated processes deliver consistent quality from prototype to high-volume manufacturing without scaling bottlenecks.
SCALE
Strategic inventory
Off-the-Shelf Availability
Strategic inventory management and flexible supply chain ensure immediate component availability, eliminating lead-time delays and enabling rapid market response.
adapt

Precision Engineering Across the Musculoskeletal Continuum
The Market We Serve
Expertise across key surgical markets, accelerating development from start to finish.
EXALTA brings private label development and precision manufacturing expertise across the full musculoskeletal spectrum, plus adjacent surgical specialties.
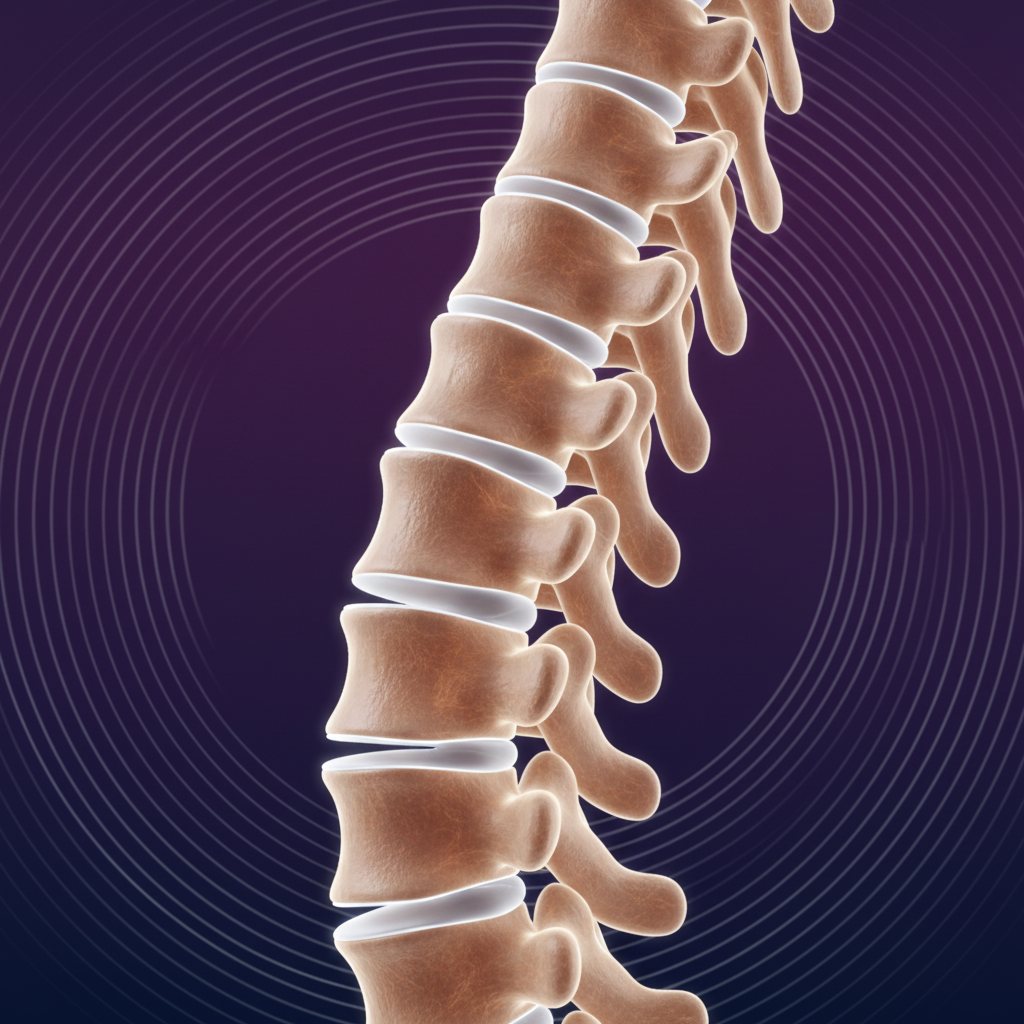
SPINE
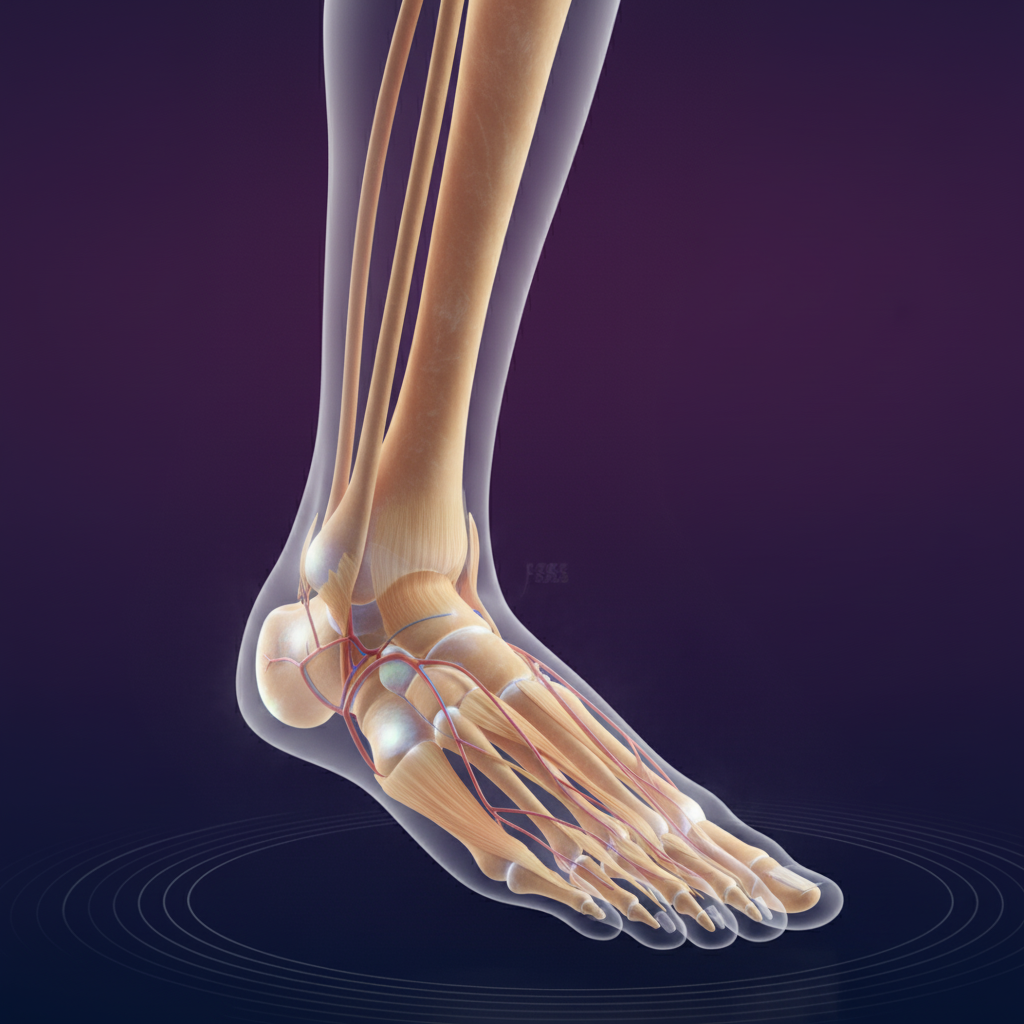
Lower Extremities
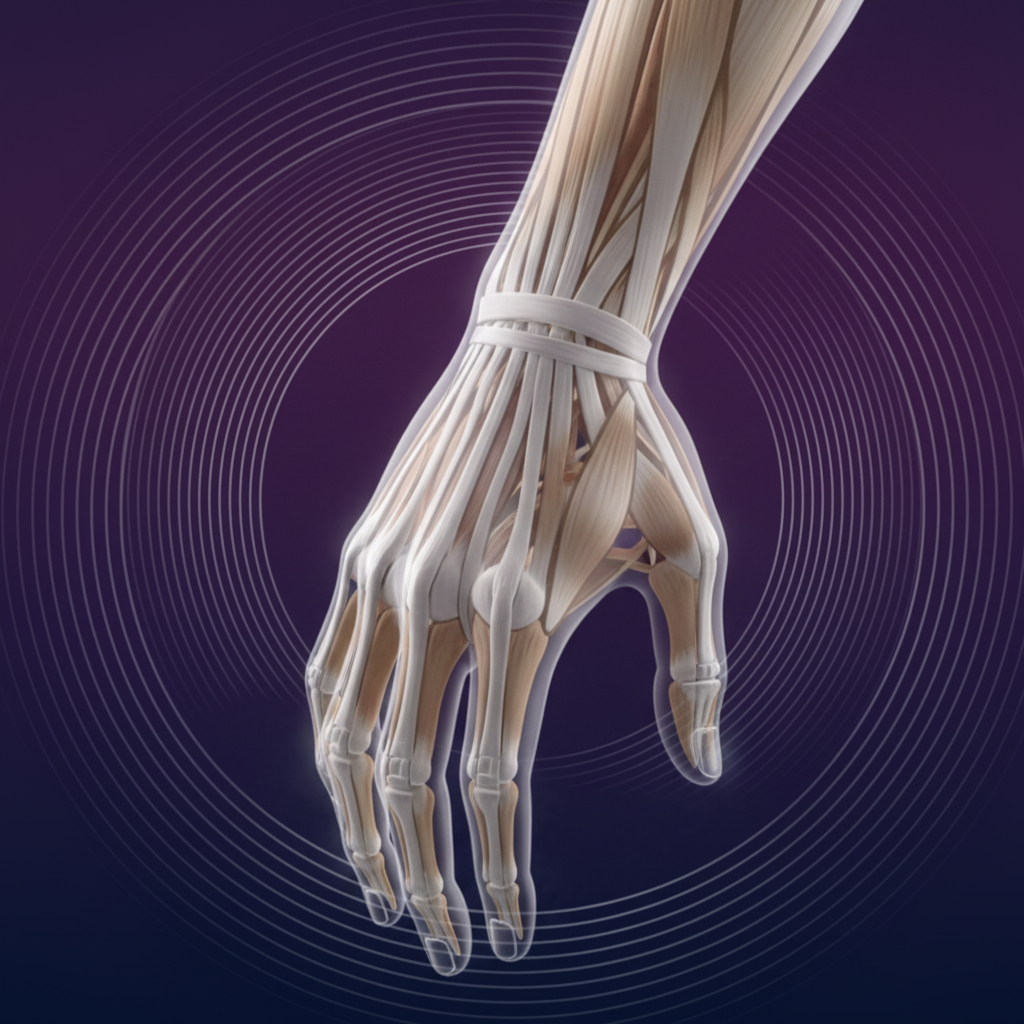
Upper Extremities
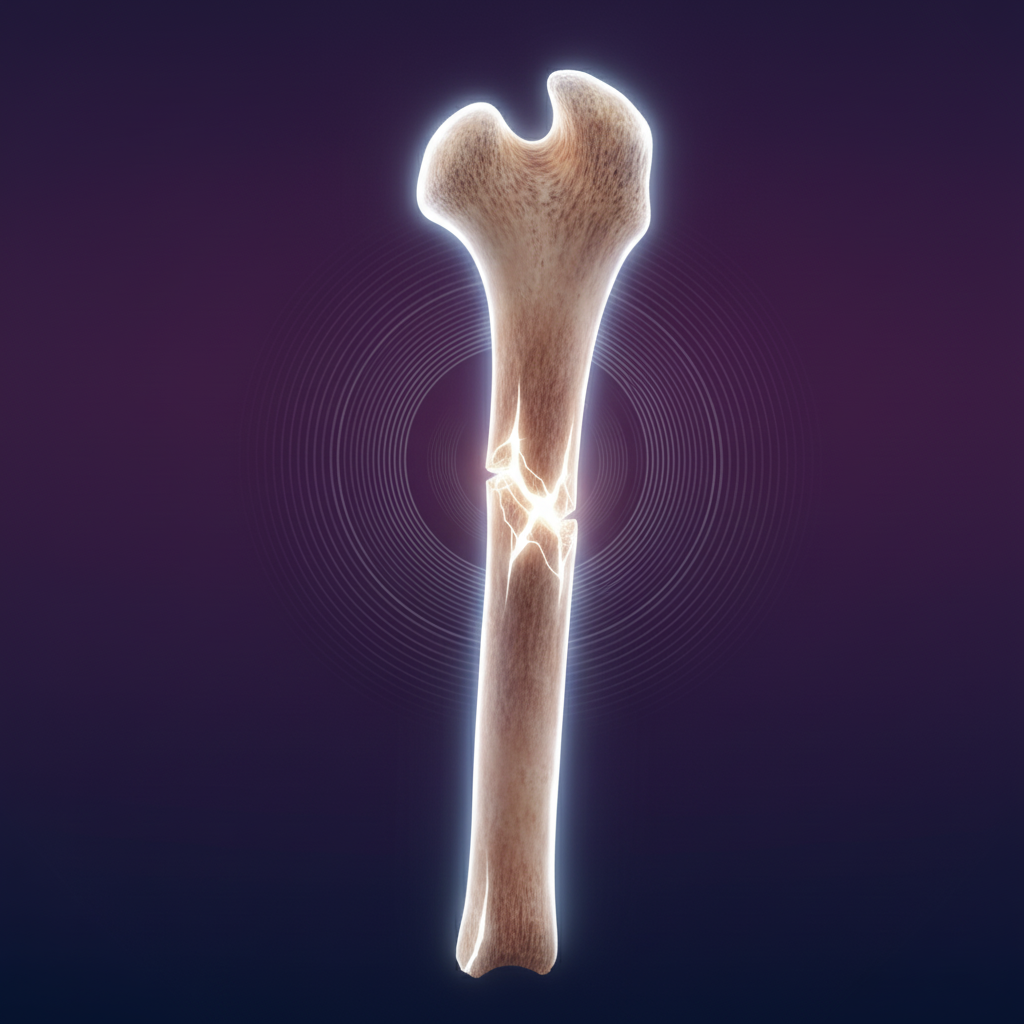
Trauma

SPINE

Lower Extremities

Upper Extremities

Trauma
Hip
Knee
Cardiothoracic

Sports Medicine

Hip

Knee

Cardiothoracic

Sports Medicine
ENT

Robotics

Navigation

Power Tools

ENT

Robotics

Navigation

